 Figuring out how the brain decides between two options is difficult. This is especially true for the human brain, whose activity is typically accessible only via the small and occasionally distorted window provided by new imaging technologies (such as functional MRI (fMRI)).
Figuring out how the brain decides between two options is difficult. This is especially true for the human brain, whose activity is typically accessible only via the small and occasionally distorted window provided by new imaging technologies (such as functional MRI (fMRI)).
In contrast, it is typically more accurate to observe monkey brains since the skull can be opened and brain activity recorded directly.
Despite this, if you were to look just at the human research, you would consider it a fact that the anterior cingulate cortex (ACC) increases its activity during response conflict. The thought is that this brain region detects that you are having trouble making decisions, and signals other brain regions to pay more attention.
If you were to only look at research with monkeys, however, you would think otherwise. No research with macaque monkeys (the ‘non-human primate’ typically used in neuroscience research) has found conflict activity in ACC.
My most recent publication looks at two possible explanations for this discrepancy: 1) Differences in methods used to study these two species, and 2) Fundamental evolutionary differences between the species.
Initially, the methods difference seems like the obvious explanation. After all, directly recording brain activity with electrodes should be more accurate than recording blood flow caused by brain activity (like with fMRI).
There’s a twist to this story, however: Measuring blood flow may actually be more accurate than recording single-unit activity in some cases.
For instance, fMRI may be more sensitive to activity in interneurons (the small neurons usually ignored by single-unit recording). As it turns out, conflict activity in ACC is thought to involve exactly these sorts of interneurons. The idea is that populations of these small cells compete for dominance between the two decision outcomes. This would cause more blood flow in ACC for fMRI to pick up (in humans), but relatively little change in the large output neurons (recorded in monkeys).
This could completely explain the discrepancy between the species, except for three things that cast doubt on it: First, local field potentials, which detect interneuron activity, yielded no results in monkeys (Emeric et al., 2008). Second, cutting out the ACC of monkeys did not change monkeys’ decision-making behaviors (Mansouri et al., 2007). Finally, single-unit recording and local field potential recording in humans undergoing surgery found conflict activity in ACC (Wang et al., 2005; Davis et al. 2005), just as with fMRI.
These findings suggest another possibility: The discrepancy is explained by a fundamental difference in the brains of the two species.
This actually makes a lot of sense, given that the two species diverged 25 million years ago. A lot of evolution has occurred in that time. If you have any doubt about that, just consider the variety of human relative to macaque monkey behaviors. Sure, we can eat, sleep, and fling feces just like monkeys, but also much much more. (Of course, a monkey could write this blog post too; it would just take much longer).
Strangely, what appears obvious to most of us is blasphemy in much of the neuroscience community (Passingham, 2009; Preuss, 2006). Most neuroscientists consider the macaque monkey brain to be essentially identical to the human brain (except, perhaps, for one or two ‘language modules’).
How could this be? Partly because there is some truth to it, and partly due to wishful thinking.
It’s true in the sense that macaque monkeys are much more similar to humans than the other often-used neuroscience animal: the rodent. Thus, relative to many other neuroscientists, monkey researchers are typically much closer to discovering the truth about the human brain.
The wishful thinking comes from all corners of the neuroscience community: We all want to know exactly what is happening in the human brain, and since we usually can’t record directly from human neurons we desperately want monkey neurons to stand in as a perfect replicas. Unfortunately, they do not.
So, all that is left to explain the discrepancy between monkey and human ACC is the possibility that there is a new brain region in human ACC that is not present in monkeys.
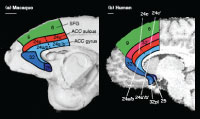
Indeed, such a region is known to exist, at least anatomically (Vogt et al. 1995; Palomero-Gallagher et al. 2008). This region, known as area 32´, is present in human (not monkey) ACC. See the above figure for the region’s location.
It is not clear exactly what this region actually does, however. We suggest that area 32´ bestows mental flexibility on humans, improving our ability to perform well on a variety of tasks by allowing us to monitor the current task difficulty and adjust our behavior to improve it. See the paper for more details.
What might this evolutionary difference mean for the future of monkey neuroscience?
It should certainly cause neuroscientists to remain skeptical of conclusions based solely on monkey research (unless, of course, they are only interested in monkey brains).
Also, it should drive researchers to directly compare human and monkey brain activity (perhaps by using fMRI with both species) to bridge findings from the two species and also to make comparisons in order to further understand our evolutionary origins.
-MC
Cole, M., Yeung, N., Freiwald, W., & Botvinick, M. (2009). Cingulate cortex: Diverging data from humans and monkeys Trends in Neurosciences, 32 (11), 566-574 DOI: 10.1016/j.tins.2009.07.001